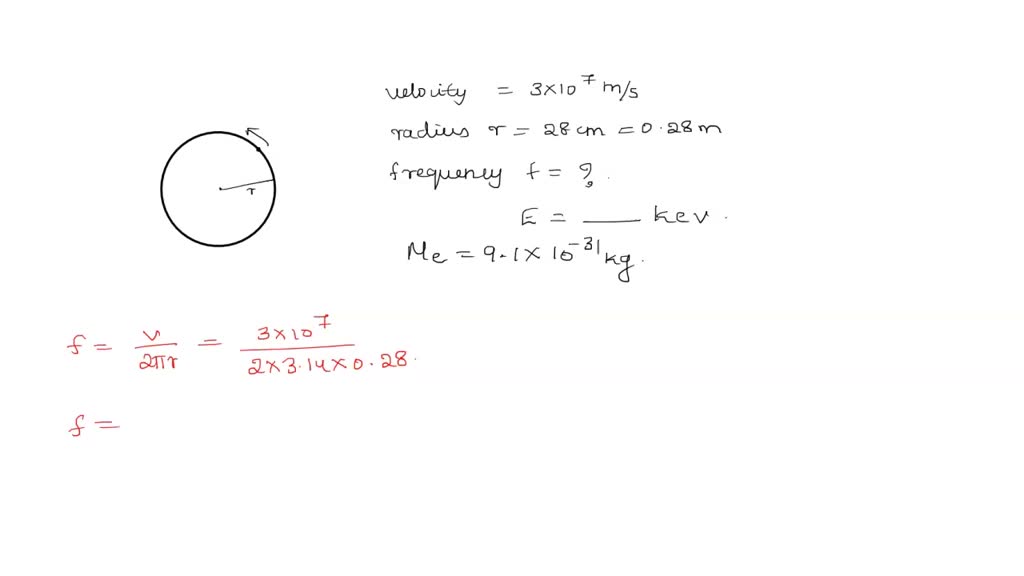
SOLVED: What is the frequency of electron which is moving with a velocity 3*10^7 m/s in the path of radius 28cm. Also calculate the energy of electron in KeV. Mass of electron=9.1*10^-31
Obtain an expression for the frequency of radiation emitted when a hydrogen atom deexcites from level n to level (n−1). - Sarthaks eConnect | Largest Online Education Community

Ratio of frequency of revolution of electron in the second excited state of He^+ and second state of hydrogen is
24/What is the frequency of revolution of electron present in 2nd Bohr's orbit of H atom? (a) 1.016 x1016 s (b) 4.065 x 1016 s (c) 1.626 x 1015 s X 1 S (d) 8.2 x 1014 s
Demonstrate that the frequency ω of a photon emerging when an electron jumps between neighbouring circular orbits - Sarthaks eConnect | Largest Online Education Community

Using bohr postulate derive the expression for the frequency of radiation emitted by an electron in hydrogen atom undergoes transition from higher energy States to lower energy state? | EduRev Class 12 Question

Calculate the angular frequency of revolution of an electron occupying the second Bohr orbit of He^(+) ion.







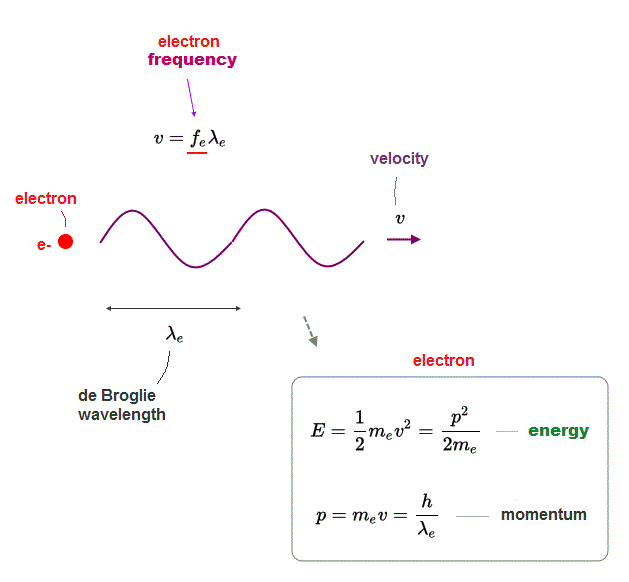



![Term 2] Derive an expression for frequency of radiation emitted when Term 2] Derive an expression for frequency of radiation emitted when](https://d1avenlh0i1xmr.cloudfront.net/135dbe2d-5958-4284-b9a2-e76dd43fdf02/slide1.jpg)